What is an acid?
An acid is a chemical which causes an aqueous solution to become
more
acidic. Another way of saying this is that an acid causes the
hydrogen ion (H+) concentration to increase. Thus
acids
are molecules which add hydrogen ions to aqueous solutions when they are
dissolved!
Where do hydrogen ions come from?
Hydrogen ions are produced when a hydrogen proton (which carries a positive charge) becomes dissociated from its electron. This produces a hydrogen ion. note: hydrogen ion = H+ = proton !
How do acids add hydrogen ions to an aqueous solution?
When an acid is added to water, the acidic hydrogen (indicated in gold) ionizes. That is the proton dissociates, leaving the electron behind. Since a hydrogen ion is produced, another common way of describing this process is that the carboxyl group ionizes. This means that the molecule now has a full negative charge.
Ionization of a carboxylic acid is more commonly depicted like this:
The fact that carboxyl
groups carry a negative charge is important in the secondary
structure of proteins, particularly Random
Coil.
Why do acids ionize?
ummm ...... I don't know ....... they just do ......... QUANTUM
MECHANICS ? ........... ask Dr. Serafin.
What is a base?
An base is a chemical which causes
an aqueous solution to become less acidic. Another way of
saying this is that an base causes the hydrogen ion (H+) concentration
to decrease. Thus bases are molecules
which remove hydrogen ions from aqueous solutions when they are dissolved!
Amino Groups are basic
The amino group is : NH2
Remember that the Nitrogen atom has 3 unpaired
electrons in its outer orbitals. ![]()
This means that nitrogen can form 3 covalent bonds. In the amino group two of these covalent bonds are with hydrogen atoms. The third covalent bond can be to any other chemical .... "R". A chemical carrying an amino group is called a primary amine.
There are two ways of depicting a primary amine as shown below:
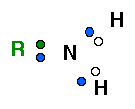 |
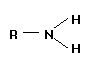 |
LEFT IMAGE: This shows the covalent bonds which are formed by sharing electrons. The 3 unpaired electrons of the nitrogen atom are shown in blue. The single electron contributed by each hydrogen atom is shown in white. The electron shared by another molecule "R" is shown in green. In all 3 covalent bonds are shown.
RIGHT IMAGE: This is the most common way of depicting a primary amine. Each of the covalent bonds represents a pair of shared electrons, but is shown simply as a line.
How do bases remove hydrogen ions from an aqueous solution?
When a base is added to water, hydrogen ions are removed from water.
Amino groups associate with hydrogen ions; this
means that the molecule now has a full positive charge.
When the amine group removes H+ from solution, the hydrogen
ion concentration decreases, and pH increases.
The fact that amino
groups carry a positive charge is important in the secondary
structure of proteins, particularly Random
Coil.
How does the pH scale work?
The pH value is defined as the negative logarithm of the hydrogen ion concentration in mol/L. The equation is:
pH = -log10[H+]The [H+] in pure water is 10^-7; therefore the pH of pure water is:
pH = -log10(10^-7) pH = - (-7) pH = 7pH 7 is often referred to as "neutral pH". Everything below pH 7 has a higher concentration of H+ and is considered acidic. Everything above pH 7 has a lower concentration of H+ and is considered basic; you can also think of this as a higher concentration of OH-.
A lower pH always means a higher concentration of H+. The biochemically useful ends of the scale are 1 M HCl, which is pH 0, and 1 M NaOH, which is pH 14. In general, cellular pH is approximately 7.2-7.4. It is very closely controlled in the cytoplasm of a healthy cell.
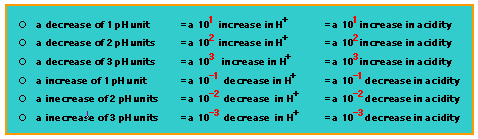
| RETURN to "Chemistry Review" Page |